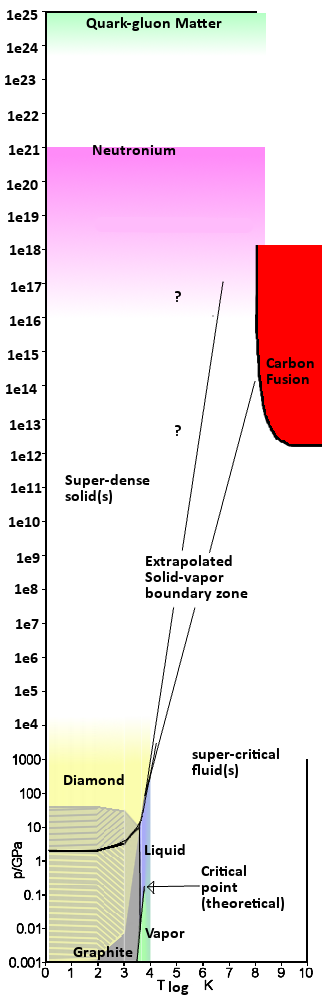
I’ve been playing around with phase diagrams, trying to figure out how exactly they work, what processes give rise to their properties, and what the boundaries look like. During this process I met with several frustrations, specifically the general lack of phase diagrams available to the public. There is some NIST software available on a demo basis, but the rest seems to be locked away behind proprietary licenses. In any case, Wikipedia proved marginally helpful and along with a few other resources I was able to get a general idea of the “normal” phases of materials.
But that only served to get me thinking… what are the limits? What’s off the edge of the maps? Super critical fluids, and exotic crystalline structures appear to be the answer… But how about even further?What about fusion, and neutronium, and all that fun stuff? Turns out it’s off the charts… so I decided to make my own. I picked Carbon for its very large range of “conventional” material states. Then I had to convert temperature to a log scale so that fusion would even fit, and then add a lot of range. After that, there was a bit of maths involving degenerate matter pressure. Dru helped a bit there, but even then the theoretical numbers are pretty hazy. So the regions are very general, and probably not accurate even within two orders of magnitude. That said, here you go.
That’s my best guess at an “extreme” matter state phase diagram. What about superfluid states, superconducting solids, etc? Don’t know. I couldn’t find even speculative numbers. But of course, at these scales, one could safely make up just about anything. No one is going to disprove it for a while now.
So, why am I thinking about this kind of stuff at all? The answer lies here.

We like solving problems about temperature and pressure. It sounds like you’ve been getting really smart about those things.